Solving the Power/Energy Paradox for High-Power Defense Applications
While often used interchangeably, power and energy are not the same. Energy is the capacity to perform and power is the rate at which that capacity is exercised — the rate at which energy is delivered. When measured, energy is an amount; power is a ratio. In relation to energy storage, a common misconception is that the larger the quantity of stored energy, the higher the potential rate of power. In fact, the opposite is true, rendering the following principles wholly counterintuitive to those unfamiliar with the nuanced mechanics of energy storage: when less energy is stored, a system is capable of producing high power and conversely, when more energy is stored, a system is capable only of producing low power. As a result, engineers are presented with a challenge in the manufacture of energy storage systems for highpower mobile applications with the highest energy density possible. In pursuit of this goal, modern research into lithium ion (Li-ion) electrochemistries is proving very promising.
Li-ion Technology
A battery is an electrochemical energy storage and conversion device, where the inter-conversion between chemical and electrical energy occurs via electron transfer reactions, or oxidation-reduction (redox) reactions. There are several classifications of batteries — primary, secondary rechargeable, mechanical replaceable, reserve and thermal. The Li-ion battery is classified as a secondary rechargeable battery. In Li-ion battery cells, the charge transfer between the positive electrode (cathode) and negative electrode (anode) is carried out by lithium ions in the electrolyte. No metallic Li is used in a Li-ion battery. Some of the commercial production cathode materials include LiCoO2, Li(NiCoAl)O2 (NCA), Li(NiCoMn)O2 (NMC), LiMn2O4 (LMO), and LiFePO4 (LFP). Currently, the most common anode material is graphite. During cell discharge, lithium ions are extracted from the anode and inserted into the cathode. The movement of Li-ion is carried out in reverse during cell charge. The electrolyte for a Li-ion battery is organic, which can be stable at voltage exceeding three volts.
In the constant search for higher power and capacity, researchers have been focusing on groups of electrode materials that can provide higher voltage and/or higher capacity. For the anode, silicon (Si) alloys — which can potentially achieve 6x the capacity of graphite — are actively pursued with the goal of overcoming material stability and cycle life issues. In the field of advanced cathode materials, higher voltage phosphates such as LiMnPO4 and LiCoPO4, as well as high voltage spinel, e.g. LiMnxNi2-xO4, are of research interest for improvement in material stability and cycleability.
Energy Density vs Power Density

• E series: high energy Li-ion
• P and M series: medium to high power
• V series: very high power; and
• U series: ultra-high power.
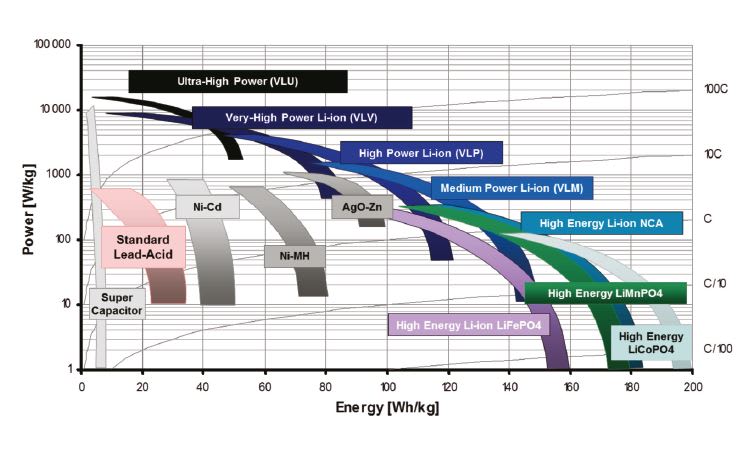
The power and energy capacities of these different battery technologies are shown in Figure 1.
For modern high-power electric weapon systems such as direct energy weapons (lasers), high-power microwave and kinetic energy weapons (electromagnetic launchers), the power requirement is very high with a very short duration (i.e. a pulsed power requirement). Traditionally, electrostatic capacitors were the preferred power sources; however, a capacitor’s power delivery is inversely exponential with respect to time. For a majority of such applications, some sort of pulse forming/shaping circuits would be required.
Recently, Saft has developed a new line of ultra-high-power batteries that have demonstrated pulsed power capability equal to or higher than that of the supercapacitor or Li-ion capacitor at a power level of tens of kW/kg. This new technology widens the choice of power sources for the electric fire community. The new pulsed power sources based on Li-ion technology have two distinct product groups: Li-ion battery and Li-ion capacitor. The Li-ion capacitor has also been termed Li-ion hybrid capacitor, because the Li-ion capacitor is composed of one Li-ion battery electrode that carries out an electrochemical reaction and one capacitor electrode that stores charges electrostatically. Figure 2 is a summarized overview of the performance specifications of each technology.
 As can be seen from the specifications, no one technology can surpass the rest in all performance categories. For example, the super-capacitor can achieve one million cycles but has the lowest power and energy density. The Li-ion battery can deliver the most current, voltage, power, and energy, but its cycle life is limited. This is due to the fact that energy charge and discharge involve Li-ion extraction and insertion into electrode materials. This action causes physical dimension change and material fatigue over cycling. Since the Li-ion capacitor is half battery, its cycle life is also limited relative to the super capacitor.
As can be seen from the specifications, no one technology can surpass the rest in all performance categories. For example, the super-capacitor can achieve one million cycles but has the lowest power and energy density. The Li-ion battery can deliver the most current, voltage, power, and energy, but its cycle life is limited. This is due to the fact that energy charge and discharge involve Li-ion extraction and insertion into electrode materials. This action causes physical dimension change and material fatigue over cycling. Since the Li-ion capacitor is half battery, its cycle life is also limited relative to the super capacitor.
By innovatively enhancing the cell’s mechanical design while optimizing the electrochemical portion of the cell, Saft SDD has successfully developed a Li-ion battery that can simultaneously supply power and energy density that is high enough to satisfy the requirements of a range of high-power defense and space applications. And while its cycle life is comparably limited, the 40,000 cycles achieved by the Saft VL5U (shown here) provides such an application with a cycle life ample enough to outlast the weapon itself.
This article was written by Yvonne Chen, Sr. Research Scientist, Saft SDD (Bagnolet, France). For more information, Click Here .
Top Stories
INSIDERDefense
![]() AI Pilots X-62A in First Aerial Dogfight - Mobility Engineering Technology
AI Pilots X-62A in First Aerial Dogfight - Mobility Engineering Technology
INSIDERAerospace
![]() Stratolaunch Approaches Hypersonic Speed in First Powered TA-1 Test Flight -...
Stratolaunch Approaches Hypersonic Speed in First Powered TA-1 Test Flight -...
INSIDERUnmanned Systems
![]() Hermeus Rolls Out First Quarterhorse Hypersonic Test Aircraft - Mobility...
Hermeus Rolls Out First Quarterhorse Hypersonic Test Aircraft - Mobility...
INSIDERManned Systems
![]() Marines Select Prototypes for Light Armored Vehicle Replacement - Mobility...
Marines Select Prototypes for Light Armored Vehicle Replacement - Mobility...
INSIDERMechanical & Fluid Systems
![]() US Navy Taps Hunt Valve for First 3D-Printed Submarine Assemblies - Mobility...
US Navy Taps Hunt Valve for First 3D-Printed Submarine Assemblies - Mobility...
INSIDERAerospace
![]() Two Prototypes Selected for Air Force Collaborative Combat Aircraft Program -...
Two Prototypes Selected for Air Force Collaborative Combat Aircraft Program -...
Webcasts
Power
![]() Transforming Transportation Manufacturing: Unlocking Growth with...
Transforming Transportation Manufacturing: Unlocking Growth with...
Automotive
![]() Accelerating the Software Development Life Cycle for the...
Accelerating the Software Development Life Cycle for the...
Automotive
![]() Applying Model-Based Design to SDV Development: A Practical...
Applying Model-Based Design to SDV Development: A Practical...
Communications
![]() Product Compliance Challenges for Wireless Radio Devices in...
Product Compliance Challenges for Wireless Radio Devices in...
Defense
![]() Building Strong Defenses: Enhancing Supply Chain Resiliency in...
Building Strong Defenses: Enhancing Supply Chain Resiliency in...